 
The ongoing challenge towards high-quality minimally invasive deep-tissue imaging calls for a new solution that combines enhancement in the spatial and temporal resolutions with a footprint reduction. Currently, minimally invasive endoscopes are widely used in neuroscience for in vivo deep brain imaging 10, 11, 12, as well as in clinical studies to assist in detecting cancers, to prescribe the right drugs and to monitor treatment response 13, 14. The numerical aperture (NA) of MM fibres approaches 0.9 8, 9, paving the way towards high-resolution but still diffraction-limited imaging. The recent emergence of spatial wavefront shaping 2, 3 has allowed a conventional step-index multimode (MM) fibre to be utilized as an ultra-thin aberration-free imaging probe 4, 5, 6, 7. Miniaturized endo-microscopy provides large depth penetration and is not limited by the interior of a hollow organ or cavity of the body. Modern microscopy demonstrates a drive towards miniaturization caused by the need to access deep tissues in vivo 1. Optical techniques have long been recognized as indispensable tools for bioimaging. The proposed approach can significantly expand the realm of the application of nanoscopy for bioimaging. We demonstrate a spatial resolution more than 2 times better than the diffraction limit and an imaging speed 20 times faster than the Nyquist limit. The new approach of super-resolution endo-microscopy does not use any specific properties of the fluorescent label, such as depletion or stochastic activation of the molecular fluorescent state, and therefore can be used for label-free imaging. We use the random nature of mode coupling in a multimode fibre, the sparsity constraint and compressive sensing reconstruction. Here, we report imaging through an ultra-thin fibre probe with a spatial resolution beyond the Abbe limit and a temporal resolution beyond the Nyquist limit simultaneously in a simple and compact setup. However, these methods typically require complicated setups and long acquisition times and are still not applicable to deep-tissue bioimaging. 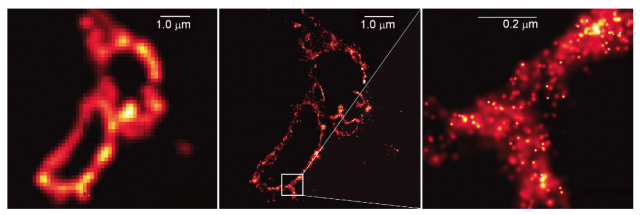
The recent development of super-resolution techniques has pushed the limits of spatial resolution. Nonetheless, far-field imaging has many limitations: the spatial resolution is controlled by the diffraction of light, and the imaging speed follows the Nyquist–Shannon sampling theorem. 
Rev.For several centuries, far-field optical microscopy has remained a key instrument in many scientific disciplines, including physical, chemical, and biomedical research. Heilemann, M., Dedecker, P., Hofkens, J., Sauer, M.: Photoswitches: key molecules for subdiffraction-resolution fluorescence imaging and molecular quantification. Piston, D.W., Kremers, G.J.: Fluorescent protein FRET: the good, the bad and the ugly. Langhorst, M.F., Schaffer, J., Goetze, B.: Structure brings clarity: structured illumination microscopy in cell biology. Gräf, R., Rietdorf, J., Zimmermann, T.: Live cell spinning disk microscopy. Image from the RCSB PDB ( of PDB ID 1EMA ( ) Ormo, M., Cubitt, A.B., Kallio, K., Gross, L.A., Tsien, R.Y., Remington, S.J.: Crystal structure of the Aequorea victoria green fluorescent protein. 44, 803–818 (1997)Ĭhudakov, D.M., Matz, M.V., Lukyanov, S., Lukyanov, K.A.: Fluorescent proteins and their applications in imaging living cells and tissues. Sheppard, C.J.R., Török, P.: Efficient calculation of electromagnetic diffraction in optical systems using a multipole expansion. Structure of the image field in an aplanatic system. Richards, B., Wolf, E.: Electromagnetic diffraction in optical systems. Sarder, P., Nehorai, A.: Deconvolution methods for 3-D fluorescence microscopy images. Academic, London (1990)īorn, M., Wolf, E.: Principles of Optics, 6th edn. Corle, R.C., Kino, G.S.: Confocal Scanning Optical Microscopy and Related Imaging Systems.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
